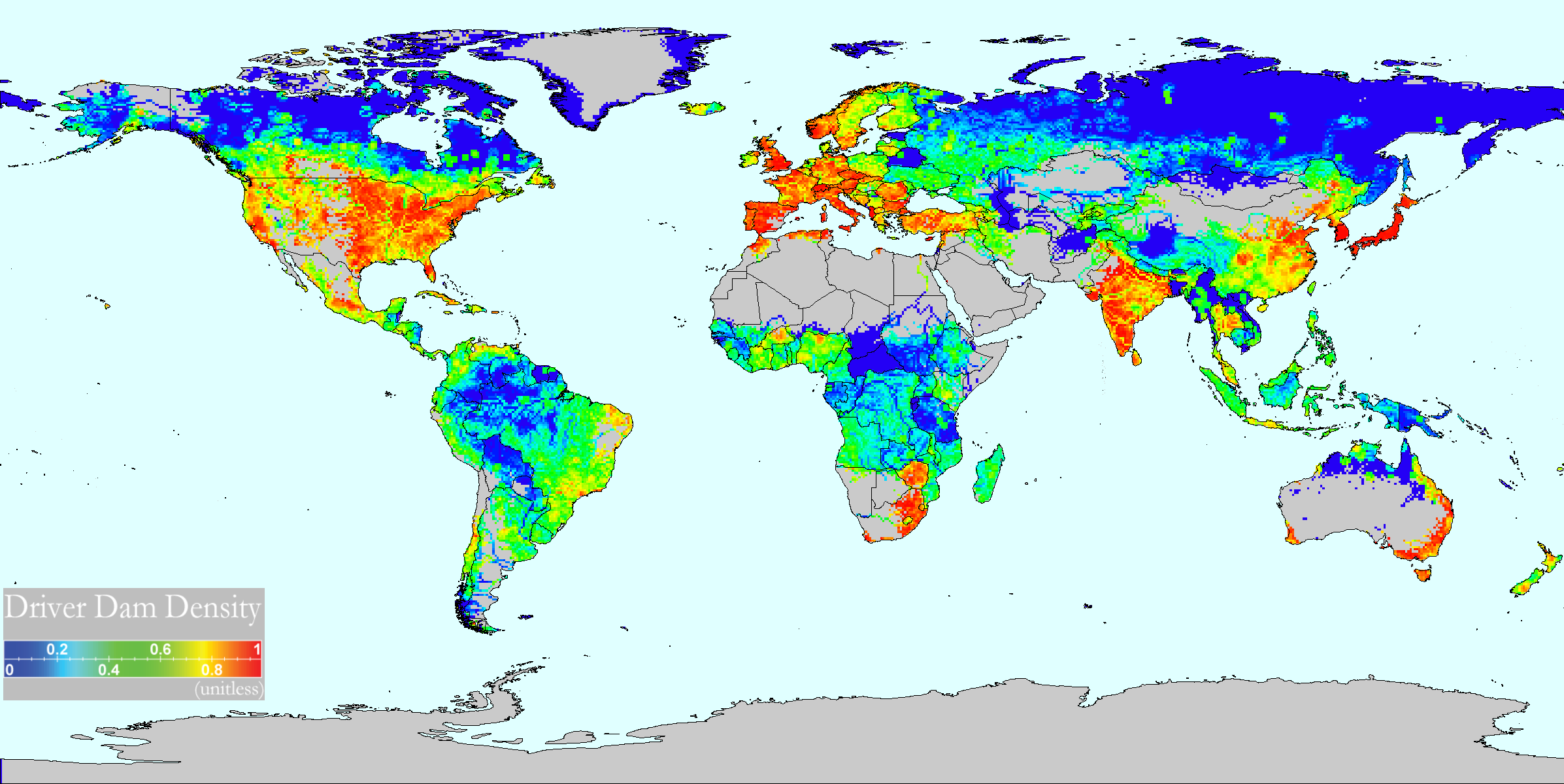
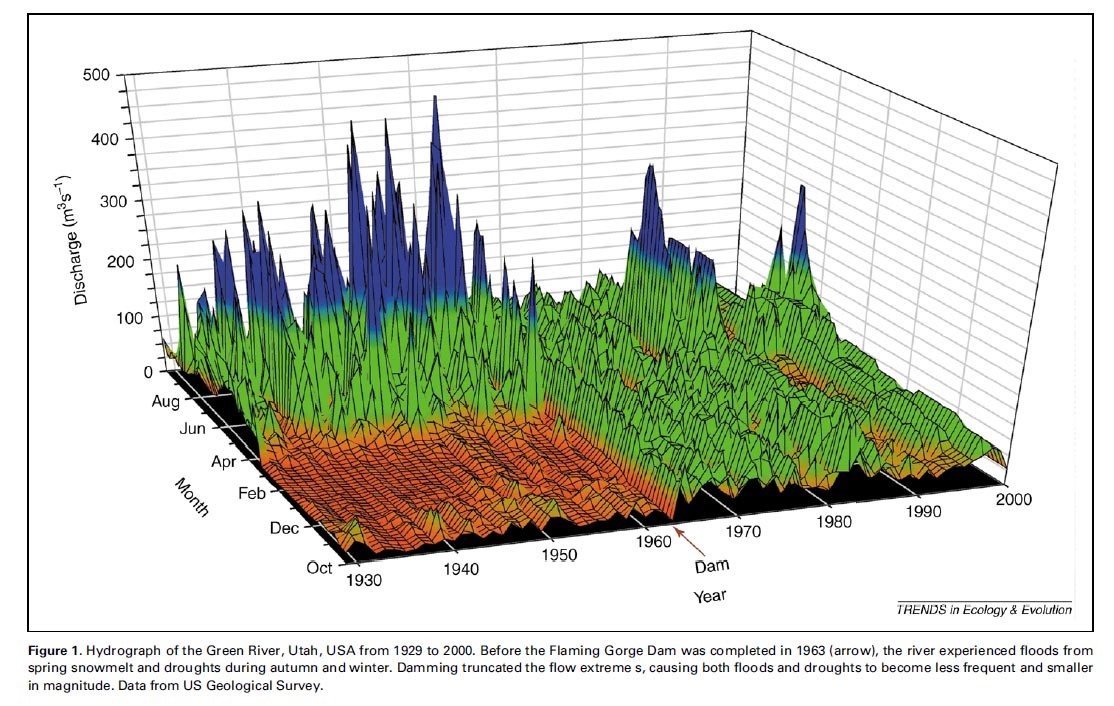
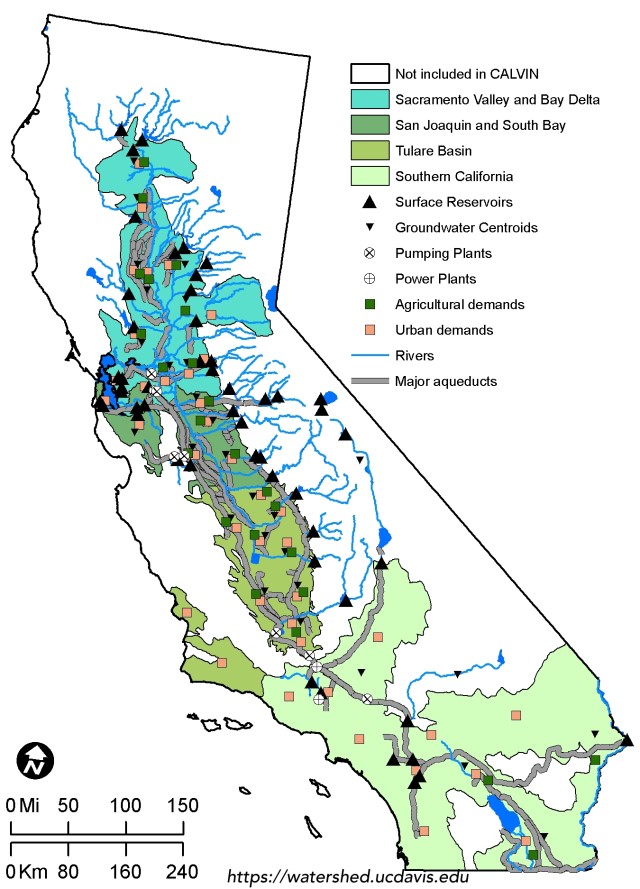
~by K. Martin Perales
Bunn, S.E., Arthington, A.H., 2002. Basic Principles and Ecological Consequences of AlteredFlow Regimes for Aquatic Biodiversity. Environmental Management 30: 492–507.Clavero, M., Hermoso, V., Aparicio, E., Godinho, F.N., 2013. Biodiversity in heavily modifiedwaterbodies: native and introduced fish in Iberian reservoirs. Freshwater Biology 58:1190–1201.
Daga, V.S., Skóra, F., Padial, A.A., Abilhoa, V., Gubiani, É.A., Vitule, J.R.S., 2015. Homogenization dynamics of the fish assemblages in Neotropical reservoirs: comparingthe roles of introduced species and their vectors. Hydrobiologia 746: 327–347.
East, A.E., Pess, G.R., Bountry, J.A., Magirl, C.S., Ritchie, A.C., Logan, J.B., Randle, T.J.,Mastin, M.C., Minear, J.T., Duda, J.J., Liermann, M.C., McHenry, M.L., Beechie, T.J.,Shafroth, P.B., 2015. Large-scale dam removal on the Elwha River, Washington, USA:River channel and floodplain geomorphic change. Geomorphology 228: 765–786.
Havel, J.E., Lee, C.E., Vander Zanden, J.M., 2005. Do reservoirs facilitate invasions into landscapes? BioScience 55: 518–525.
Johnson, P.T., Olden, J.D., Vander Zanden, M.J., 2008. Dam invaders: impoundments facilitatebiological invasions into freshwaters. Frontiers in Ecology and the Environment 6: 357–363.
Kanehl, P.D., Lyons, J., Nelson, J.E., 1997. Changes in the Habitat and Fish Community of theMilwaukee River, Wisconsin, Following Removal of the Woolen Mills Dam. North American Journal of Fisheries Management 17: 387–400.
Marchetti, M.P., Moyle, P.B., 2001. Effects of Flow Regime on Fish Assemblages in a Regulated California Stream. Ecological Applications 11: 530–539.
Marchetti, M.P., Light, T., Moyle, P.B., Viers, J.H., 2004. Fish invasions in California watersheds: testing hypotheses using landscape patterns. Ecological Applications 14:1507–1525.
Moyle PB 2002. Inland Fishes of California. Berkeley: University of California Press.O’Hanley, J.R., 2011. Open rivers: Barrier removal planning and the restoration of free-flowing rivers. Journal of Environmental Management 92: 3112–3120.
Rahel, F.J., 2002. Homogenization of Freshwater Faunas. Annual Review of Ecology and Systematics 33: 291–315.
Ricciardi, A., 2006. Patterns of invasion in the Laurentian Great Lakes in relation to changes in vector activity. Diversity and Distributions 12: 425–433.
Stromberg JC, S J Lite, R Marler, C Paradzick, PB Shafroth, D Shorrock, JM White, & MSWhite. 2007. Altered stream flow regimes and invasive plant species: the Tamarix case. Global Ecology and Biogeography 16: 381–393.
Taylor, B. & Ganf, G.G. (2005) Comparative ecology of two co-occurring floodplain plants: thenative Sporobolus mitchellii and the exotic Phyla canescens. Marine and Freshwater Research, 56: 431–440.
Vondracek, B., Baltz, D.M., Brown, L.R., Moyle, P.B., 1989. Spatial, seasonal and dieldistribution of fishes in a California reservoir dominated by native fishes. Fisheries
Research 7: 31–53.
Vörösmarty, C.J., McIntyre, P.B., Gessner, M.O., Dudgeon, D., Prusevich, A., Green, P.,Glidden, S., Bunn, S.E., Sullivan, C.A., Liermann, C.R., Davies, P.M., 2010.
Globalthreats to human water security and river biodiversity. Nature 467: 555–561.Walther, G.-R., Roques, A., Hulme, P.E., Sykes, M.T., Pyšek, P., Kühn, I., Zobel, M., Bacher, S., Botta-Dukát, Z., Bugmann, H., others, 2009. Alien species in a warmer world: risks and opportunities. Trends in ecology & evolution 24: 686–693.