Adaptation to ecological novelty: Can species keep pace?
“…I’m simply saying that life… finds a way.” -Dr. Ian Malcolm, Jurassic Park (1993)
Novelty is not new to life on Earth. Organisms have been adapting to novel challenges for millennia. But the key difference between adaptation over the evolutionary history of Earth and adaptation in recent history is the pace of change organisms have had to keep up with.
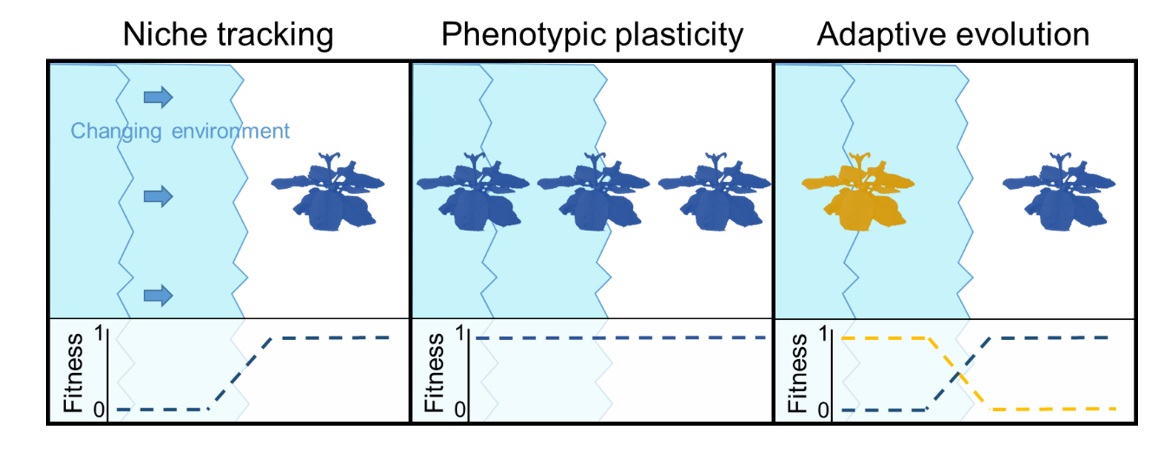
Populations can adapt to novel conditions by three principle means (Figure 1): 1) niche tracking, 2) phenotypic plasticity, and 3) adaptive evolution.
Figure 1. Modes of adaptation:
- Niche tracking involves avoiding the new environment, and could be generalized to any behavior that moderates the effect of a novel environment on fitness.
- Phenotypic plasticity occurs when phenotypic expression is flexible and can maintain fitness in either environment.
- Adaptive evolution involves changes in allele frequencies at loci that result in increased fitness in the new environment, often at the expense of fitness in the original environment.
- Note: Niche tracking and phenotypic plasticity do not involve any evolutionary change, whereas adaptive evolution arises when populations become locally adapted to the new environment.
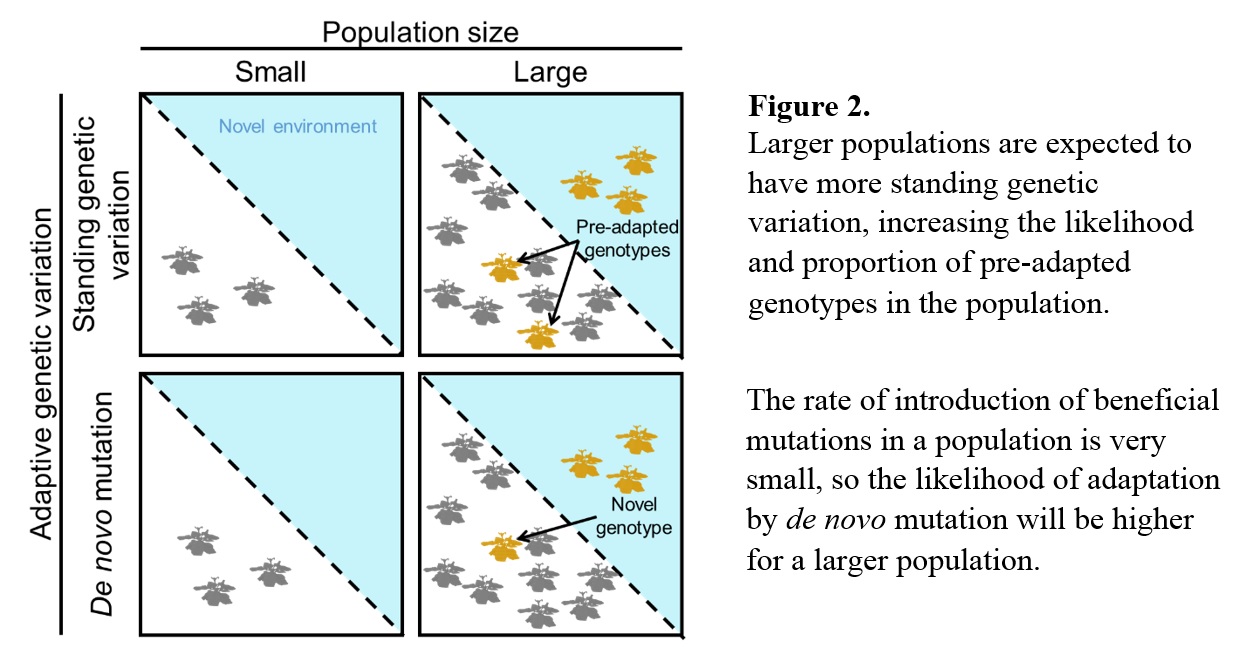
The ability to genetically adapt hinges on one thing: the availability of standing genetic variation on which natural selection can act. Adaptation over evolutionary time scales (thousands to millions of years) can occur by the generation of beneficial traits by gradual genomic changes ranging from whole genome duplications to point mutations. But adaptation on ecological time scales (tens to hundreds of years) requires adaptive genetic variation to already be available prior to the environmental shift, clearly presenting a challenge for adaptation to novelty on ecological timescales. This challenge can be overcome by species with large population sizes and short generation times, such as some arthropods, marine species, microbes, and plants, because adaptive mutations arise more frequently, and are less likely to be lost due to demographic changes (Figure 2). But many species, particularly those of conservation concern, will not have the genetic capacity to keep pace with ecological change.
Despite the rapid pace of changes in biotic and abiotic factors, many contemporary evolutionary success stories have emerged. A significant proportion of these stories involve species that are considered invasive or pests, but many less negatively-charged examples also exist. For example:
- Eurasian blackcaps have exhibited genetic changes in migratory behavior to utilize more northerly wintering habitat (Berthold et al. 1992) while great tits have evolved greater plasticity of reproductive timing to better synchronize with changing seasonal food availability (Nussey et al. 2005);
- Peppered moths and field mice have rapidly evolved color polymorphisms to avoid predation in novel environments (Grant & Wiseman 2002, Linnen et al. 2009)
- Marine phytoplankton evolved higher heat tolerances under experimental warming conditions (Listmann et al. 2016) while rapid evolution of salt-tolerance in marine zooplankton has facilitated expansion into freshwater habitats (Lee 2016);
- Several plant species have rapidly evolved heavy-metal tolerance, enabling colonization of polluted mine tailings (Wu et al. 1975, Baker et al. 1986).
Among drivers of rapid change, climate change is particularly concerning, as the majority of adaptive responses thus far have not involved genetic changes in thermal physiology, but rather, thermal niche tracking (Bradshaw & Holzapfel 2006). If trends continue, species with long generation times, poor dispersal abilities, and small population sizes may not be able to adapt to warming temperatures, particularly in the tropics (Parmesan 2006, Huey et al. 2012), and cold-adapted species may run out of niche space to track. Life as we know it may not always be able to find a way.
In addition to the challenge of the pace of contemporary environmental change, overall loss of habitat in human-dominated landscapes has exerted downward pressure on population sizes, making adaptive evolution in many species highly unlikely. For these species, persistence may require substantial conservation effort. Facilitated migration and habitat conservation and restoration may buy species time to evolve. Controlled breeding may help maintain effective population sizes, though some have raised concerns over the potential for outbreeding depression (Frankham 2010), and some species may already have crossed the point of no return.
More controversial actions such as facilitating “adaptive introgression” (Hamilton & Miller 2016), the genetic exchange from adapted to maladapted species, may be necessary to increase standing genetic variation for adaptive evolution. Much excitement now surrounds emergent technologies once relegated to science fiction novels, such as genome editing facilitated by CRISPR/Cas9, which could enable direct insertion of adaptive alleles into struggling populations. However, we must be cautious of “techno-arrogance” that addresses symptoms while ignoring causes of species declines, and ultimately promises more than it can deliver (Meffe 1993).
On a hopeful note, many species likely already possess the standing genetic variation necessary to maintain fitness in the increasingly novel environmental space they occupy. Whether these are the species we want to be the evolutionary “winners” in the era of ecological novelty, however, remains an open question. Life may yet find a way, but the fate of some species may ultimately be in our hands.
~By Michael Crossley~
References
Baker et al. (1986) Induction and loss of cadmium tolerance in Holcus lanatus L. and other grasses. New Phytologist 102, 575-587.
Berthold et al. (1992) Rapid microevolution of migratory behavior in a wild bird species. Nature 360, 668-670.
Bradshaw & Holzapfel (2006) Evolutionary response to rapid climate change. Science 312, 1477-1478.
Frankham (2010) Challenges and opportunities of genetic approaches to biological conservation. Biological Conservation 143, 1919-1927.
Grant & Wiseman (2002) Recent history of melanism in American peppered moths. Journal of Heredity 93, 86-90.
Hamilton & Miller (2016) Adaptive introgression as a resource for management and genetic conservation in a changing climate. Conservation Biology 30, 33-41.
Huey et al. (2012) Predicting organismal vulnerability to climate warming: roles of behavior physiology and adaptation. Philosophical Transactions of the Royal Society of Britain 367, 1665-1679.
Lee (2016) Evolutionary mechanisms of habitat invasions, using the copepod Eurytemora affinis as a model system. Evolutionary Applications 9, 248-270.
Linnen et al. (2009) On the origin and spread of an adaptive allele in deer mice. Science 325, 1095-1098.
Listmann et al. (2016) Swift thermal reaction norm evolution in a key marine phytoplankton species. Evolutionary Applications (in press)
Meffe GK (1993) Techno-arrogance and halfway technologies: Salmon hatcheries on the Pacific Coast of North America. Conservation Biology 6, 350-354.
Nussey et al. (2005) Selection on heritable phenotypic plasticity in a wild bird population. Science 310, 304-306.
Parmesan (2006) Ecological and evolutionary responses to recent climate change. Annual Reviews of Ecology, Evolution, and Systematics 37, 637-669.
Wu et al. (1975) The potential for evolution of heavy metal tolerance in plants. III. The rapid evolution of copper tolerance in Agrostis tenuis. Heredity 34, 165-187.